Prostate Cancer Outcomes: An International Registry to Improve Outcomes in Men with Advanced Prostate Cancer
Type of Study: Epidemiological
Sponsor / Support: LACOG, Movember
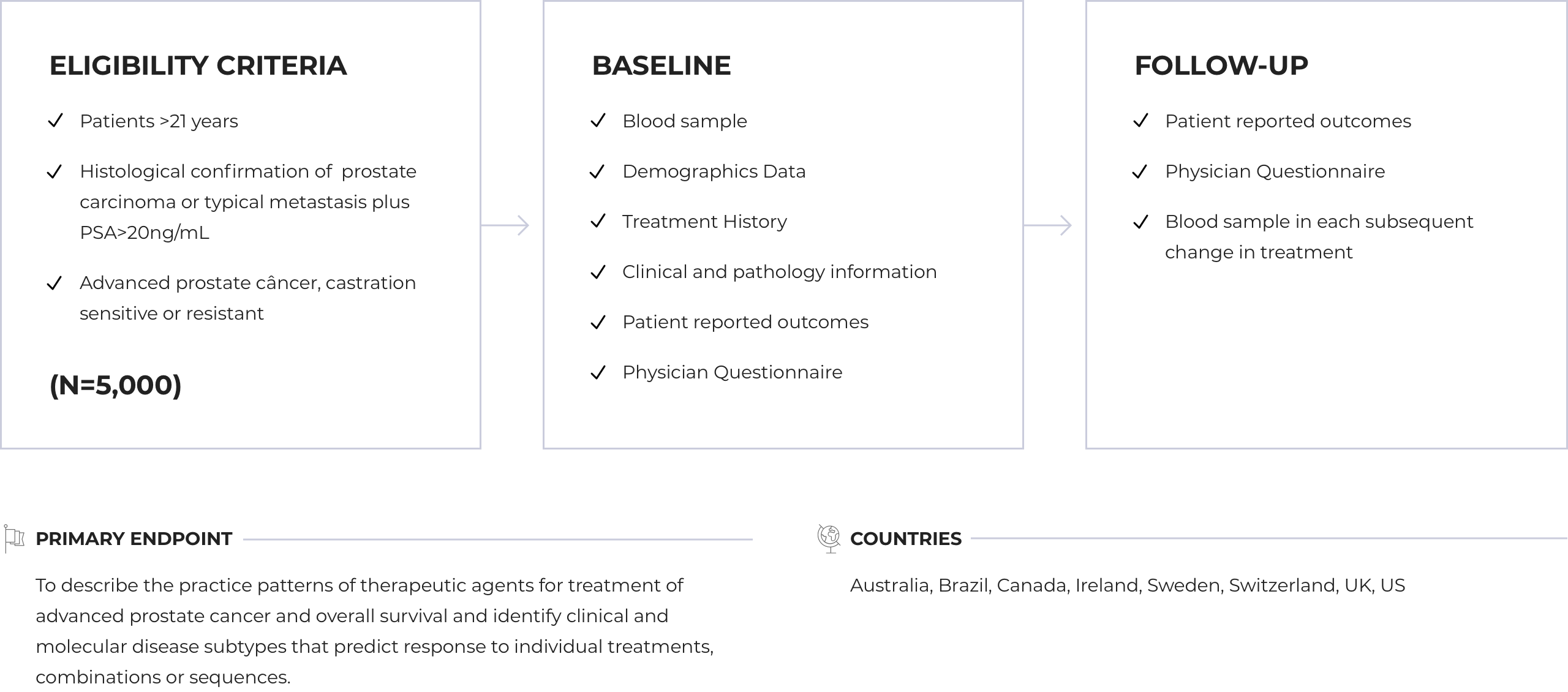
Primary Objectives: Describe the practice patterns of therapeutic agents for treatment of advanced prostate cancer and overall survival and identify clinical and molecular disease subtypes that predict response to individual treatments, combinations or sequences.
Sample Size: 5.000 patients
Principal Investigator: Andre Fay
Countries LATAM: Brazil
Clinicaltrials.gov Identifier: NCT03151629
The study is open to patients participation in the following research sites:
Ongoing studies
Breast
GBECAM 0115 – AMAZONA III
LACOG 0115 – LORELEI
LACOG 0221 – BRAVE
LACOG 0413 – Male Breast Cancer
LACOG 0419 – NEOSAMBA
LACOG 0615 – LATINABreast
LACOG 0715 – PALLAS
LACOG 0721 – Cherry Pick
LACOG 2118 – ALEXANDRA/IMpassion 030
Gastrointestinal
LACOG/GTG 0119 – NET
LACOG 0222 – GASPAR
LACOG 0421 – ACTION HIV
LACOG/GTG 1318 – Anal Registry
Genitourinary
LACOG 0121 – PET-PSMA
LACOG 0217 – IRONMAN
LACOG 0218 – Hercules
LACOG 0519 – PEACE III
LACOG 0522 – DORA
LACOG 0620 – ExBAT
LACOG 1120 – RENAL REGISTRY
LACOG 1818 – Prostate Cancer Registry
LACOG 2018 – Foundation Penile
Gynecological
EVA/LACOG 0123 | GOG-3043 – ROCC Trial
LACOG 0223 / GOG-3073 – ROSELLA
LACOG 0521 – MADONNA
LACOG 0623 – eVOLVE-Cervical
LACOG 0820 – EVITA LATAM
LACOG 0920 – Senticol III
LACOG 1018 – PALBO in Ovarian Cancer
LACOG 1220 – CONOR
Head and Neck
LACOG 0319
LACOG 0720 – SMART-KEY
Lung
LACOG 0116 – LATINO LUNG
LACOG 0118 – RELANCE
LACOG 0120 – EAP durva lung cancer
LACOG 0322 – EDUR-BRA
LACOG 0821
LACOG 1918
LACOG 2218 – PACIFIC BRAZIL
Radiation
LACOG 0122 – RADIANT
Closed studies
Breast
LACOG 0111 – MGH
LACOG 0312 – METASTATIC BC
LACOG 0414
LACOG 0801 – GLICO
Genitourinary
LACOG 0415 – APA in Prostate Cancer
LACOG 0515 – Testicular Registry
LACOG 1518 – Bladder Cancer Registry
Gynecological
LACOG 0215 – EVITA
Head and Neck
LACOG 0318 – Induction
Lung
LACOG 0211 – ALK
LACOG 0417 – CNS MTX
LACOG 0618
Parsimony
Neuro
LACOG 0619
Other studies
LACOG 0213 – AUGMENT
LACOG 0214 – FINEP